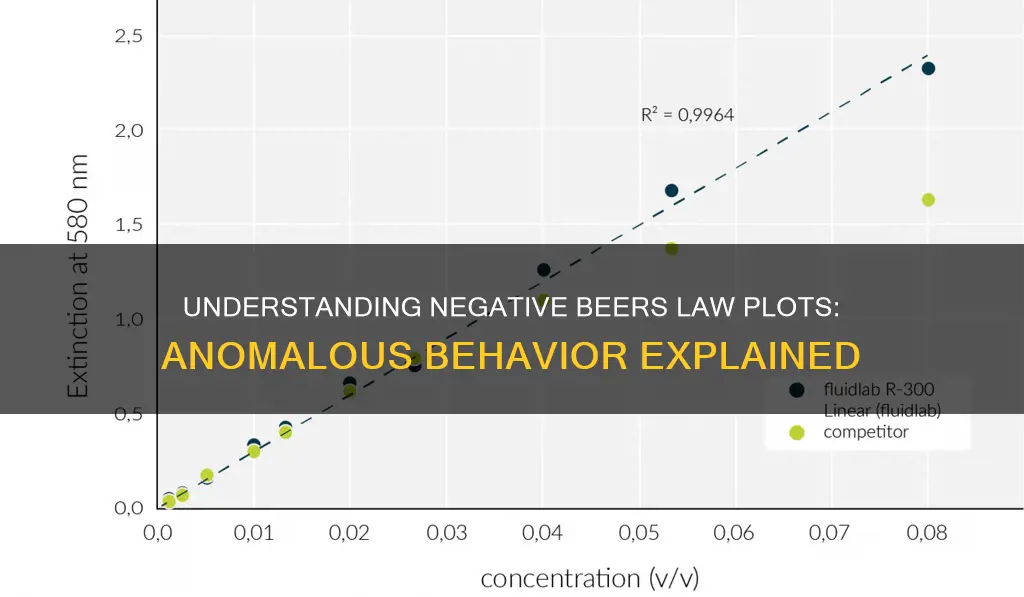
Beer's Law is a simple linear proportionality between concentration and absorbance. The law indicates that there is a linear relationship between the two variables, and by plotting a calibration curve of solutions of known concentration and their respective absorbance, the concentration of an unknown sample can be determined. However, the plot of the data may not always be linear and can deviate from Beer's Law, indicating that the standards were improperly prepared or that there is an unknown interference in the sample. This deviation can also be caused by the presence of stray radiation at higher concentrations, resulting in a negative deviation from Beer's Law. Additionally, the quality of the spectrophotometer and the material of the cuvette can impact the accuracy of the absorbance measurements.
| Characteristics | Values |
|---|---|
| Relationship | Linear proportionality between concentration and absorbance |
| Ideal Plot | Straight line |
| Slope of the Plot | Extinction Coefficient |
| Deviation from Beer's Law | Caused by the presence of stray radiation at higher concentrations |
| Calibration | Absorbances of standard samples are measured and plotted on the y-axis, with known analyte concentrations on the x-axis |
What You'll Learn

The curvature at higher concentrations
Beer's Law, or the Beer-Lambert Law, is a simple linear proportionality between concentration and absorbance. The ideal plot of Beer's Law is a straight line with a y-intercept of zero.
However, at higher concentrations, the Beer's Law plot may show curvature, or non-linearity, which represents a negative deviation from the ideal plot. This curvature is caused by the presence of stray radiation and the increased likelihood of interactions between sample molecules at higher concentrations. As a result, the assumption used to derive Beer's Law breaks down, leading to a negative deviation from the law.
At higher concentrations, a relatively small change in transmittance can lead to a large change in absorbance. This is because the point where only 10% of the radiation is transmitted through the sample corresponds to an absorbance value of 1. An absorbance of 1 represents a decrease in light intensity of 90%, while an absorbance of 2 represents a decrease in light intensity of 99%. Therefore, even a small change in transmittance can have a significant impact on the absorbance value at high concentrations.
Federal Law Overturned: Is It Possible?
You may want to see also

Poor solubility of the compound in the solvent
Beer's Law, also known as the Beer-Lambert Law, establishes a linear relationship between concentration and absorbance. By plotting the absorbance of a set of different concentrations of a compound, one can determine whether the relationship holds. The ideal plot is a straight line that goes through the origin.
However, a Beers Law plot can decrease due to various factors, one of which is the poor solubility of the compound in the solvent. When a compound has poor solubility in the chosen solvent, it may form a turbid solution at the concentrations being tested. This turbidity can scatter light, leading to inaccurate absorbance readings and deviations from the expected linear relationship.
To address this issue, one can consider the following strategies:
- Solvent Selection: Choose a solvent in which the compound is known to have better solubility. This may involve researching solvent-solute compatibility or conducting preliminary solubility tests.
- Temperature Modification: Increasing the temperature of the solvent can sometimes enhance the solubility of the compound. This approach, known as "heating," can help improve solubility without changing the solvent.
- Ultrasonic Treatment: Applying ultrasonic energy to the solvent can create cavitation bubbles, which may temporarily increase the solubility of the compound, thus improving its solubility during the testing process.
- Stirring and Agitation: Mechanical agitation or stirring can promote solubility by ensuring continuous contact between the compound and the solvent, enhancing dissolution.
- Solvent Mixtures: In some cases, creating a mixture of two or more solvents may improve the compound's solubility. This approach can be particularly useful when a single solvent does not provide adequate solubility.
By employing these strategies, one can mitigate the issues arising from poor compound solubility and improve the accuracy of the Beer's Law plot. It is important to note that the choice of strategy will depend on the specific compounds and solvents involved, as well as the experimental conditions.
Introducing Bills: Who Has the Power?
You may want to see also

Quality of the spectrophotometer
The quality of the spectrophotometer is a critical factor in obtaining accurate and reliable results when working with Beer's Law. Beer's Law, also known as the Beer-Lambert Law, is a fundamental principle in optical spectroscopy, describing the relationship between the attenuation of light and the properties of the material through which it travels.
The spectrophotometer plays a pivotal role in measuring light intensity and absorbance. An ideal Beer's Law plot should exhibit a linear relationship between absorbance and concentration, with the plot passing through the origin. However, limitations in the quality of the spectrophotometer can introduce deviations from this ideal linear relationship.
One key aspect of spectrophotometer quality is its ability to accurately measure low light intensities. As absorbance increases, representing a greater decrease in light intensity, the quality of the instrument becomes more critical. For example, an absorbance of 1 represents a 90% decrease in light intensity, while an absorbance of 2 corresponds to a 99% decrease. The useful range of absorbance measurements is influenced by the spectrophotometer's sensitivity and precision in measuring low light levels.
Additionally, the choice of cuvettes used in the spectrophotometer is essential. For ultraviolet measurements, quartz cuvettes are recommended because cheaper glass or disposable plastic cuvettes are opaque to UV light. The material and thickness of the cuvettes can introduce deviations from Beer's Law, affecting the accuracy of your results.
Furthermore, it is important to consider the limitations of Beer's Law itself when assessing the quality of a spectrophotometer. Beer's Law assumes a simple linear relationship between concentration and absorbance. However, deviations from this linearity often occur at higher concentrations due to changes in the absorbing species or the bulk solution. These deviations can lead to negative deviations from Beer's Law, such as curvature in the plot. Therefore, it is crucial to understand the concentration range over which Beer's Law is applicable for a given system.
In conclusion, the quality of the spectrophotometer is a critical factor in obtaining reliable results when working with Beer's Law. The instrument's ability to accurately measure low light intensities, the choice of cuvettes, and an understanding of the limitations of Beer's Law itself are all essential aspects of ensuring the quality and accuracy of your measurements.
Common-Law Marriage: Tax Implications and Claims
You may want to see also

The material of the cuvette
Quartz cuvettes are ideal for ultraviolet (UV) measurements. This is because cheaper glass or disposable plastic cuvettes are often opaque to UV light, meaning they do not allow UV light to pass through. Using the wrong type of cuvette for UV measurements can lead to inaccurate results. Therefore, it is crucial to select the appropriate cuvette material for the specific type of light being used in the experiment.
The length of the cuvette, also known as the path length, is another important consideration. The path length is directly proportional to the absorbance of the sample. In other words, the longer the cuvette, the more light will be absorbed by the sample. This is an essential factor to keep in mind when designing experiments and interpreting results. The path length is also related to the concentration of the sample, as longer path lengths can accommodate more concentrated solutions.
Additionally, the quality of the cuvette can impact the accuracy of measurements. Cuvettes should be clean and free of scratches or impurities that could affect the light transmission. It is also important to consider the refractive index of the cuvette material, especially when working with high-precision measurements. The refractive index of the cuvette should match the refractive index of the sample as closely as possible to minimize errors.
In summary, the material of the cuvette plays a significant role in the accuracy of measurements when using Beer's Law. Researchers should carefully select the appropriate cuvette material, considering factors such as the type of light being used, the path length, and the quality of the cuvette, to ensure reliable results. By choosing the right cuvette, deviations from Beer's Law due to factors such as stray radiation and improper sample preparation can be minimized.
Elder Law Attorneys: Guaranteed Approval?
You may want to see also

Unknown interference in the sample
Beer's Law, also known as the Beer-Lambert Law, is a simple linear proportionality between concentration and absorbance. The ideal plot of the data should be a straight line that goes through the origin. If the plot is not linear or if the y-intercept deviates from the origin, it could be due to unknown interference in the sample.
Firstly, the choice of cuvette material can play a role. For ultraviolet measurements, quartz cuvettes are recommended because cheaper glass or disposable plastic cuvettes may be opaque to UV light, affecting the light transmission and absorbance measurements.
Secondly, the compound's solubility in the solvent can be a factor. If the compound has poor solubility, it may form a turbid solution at the tested concentrations, deviating from Beer's Law.
Additionally, the presence of stray radiation can cause curvature at higher concentrations, leading to negative deviations from Beer's Law. This is because the sample molecules are more likely to interact at higher concentrations, breaking down the assumptions used to derive the law.
Furthermore, the limitations of the spectrophotometer can impact the results. While it is not a general rule, absorbances above 1 may be unreliable due to the instrument's ability to accurately measure low light intensities. An absorbance of 1 represents a 90% decrease in light intensity, and as absorbance values increase, small changes in transmittance can lead to large changes in absorbance at high concentrations.
To address these potential interferences, scientists can take several approaches. Diluting the sample can help bring the data into the linear portion of the curve if non-linearity occurs at absorbance values higher than one. Alternatively, using a non-linear higher-order equation may be acceptable for calculating concentrations if non-linearity occurs at absorbance values lower than one.
Studying Law in the USA: Options for Indians
You may want to see also
Frequently asked questions
Beer's Law is a simple linear proportionality between concentration and absorbance. It indicates that there is a linear relationship between absorbance and concentration.
A decrease in a Beer's Law plot indicates a negative deviation from Beer's Law. This can be caused by the presence of stray radiation at higher concentrations. It could also be due to the compound's poor solubility in the solvent, which may form a turbid solution at the concentrations being tested.
The accuracy of a Beer's Law plot can be influenced by the quality of the spectrophotometer, the material of the cuvette, and the preparation of the standards. Quartz cuvettes, for example, are recommended for ultraviolet measurements as they allow UV light to pass through, unlike cheaper glass or plastic alternatives.