The rate law is an important concept in chemistry that helps us understand the relationship between the rate of a chemical reaction and the concentrations of its reactants. When studying a chemical reaction, various factors come into play, such as the chemical properties of the reactants, the conditions under which the reaction occurs, the mechanism, the rate, and the equilibrium it reaches. One of the key factors influencing the rate of a reaction is the presence of a catalyst, which can speed up the process. While catalysts are not included in the balanced overall chemical equation, they can still impact the reaction rate. This raises the question: can a catalyst appear in the rate law?
What You'll Learn
- Catalysts can be included in a reaction without being in a rate equation
- The rate equation is more concerned with establishing equilibrium conditions
- The rate law is experimentally determined
- The rate equation is based on empirical data
- The concentration of active particles on a catalyst's surface should be included in the rate equation

Catalysts can be included in a reaction without being in a rate equation
A catalyst is a substance that speeds up a chemical reaction or lowers the temperature or pressure required to start one. It achieves this by lowering the activation energy, which is the energy barrier that must be overcome for a chemical reaction to occur. Catalysts are not consumed during the reaction and remain chemically unchanged at its end.
The rate law is experimentally determined and can be used to predict the relationship between the rate of a reaction and the concentrations of reactants and products. According to the law of mass action, the rate of a chemical reaction at a constant temperature depends only on the concentrations of the substances that influence the rate. While catalysts can influence the reaction rate, they do not appear in the balanced overall chemical equation. This is because the rate equation is often more concerned with establishing equilibrium conditions than with determining the time it takes to reach equilibrium.
The rate equation is generally created on an empirical basis, and the dependence on catalyst concentration is usually complex and not needed. In most non-enzymatic reactions, the catalyst concentration is less than that of any of the reactants. However, in enzymatic reactions, such as the enzyme catalase converting H2O2, the enzyme has a greater concentration than H2O2 under normal conditions.
Heterogeneous catalysts, which are not part of the reaction mixture, are usually not included in the rate equation as their concentration in the mixture is zero by definition. However, the concentration of active particles on the surface of the heterogeneous catalyst should be included in the rate equation. While the presence of a catalyst can significantly impact the rate of a reaction, it may not always be reflected in the rate equation due to the complex and multifaceted nature of chemical reactions.
Benelli's Law Enforcement Guns: Who Can Purchase Them?
You may want to see also

The rate equation is more concerned with establishing equilibrium conditions
The rate equation is a fundamental concept in chemistry, helping us understand the intricate dynamics of chemical reactions. While it plays a crucial role in deciphering reaction rates, its significance extends beyond that. Indeed, the rate equation is often more concerned with establishing equilibrium conditions, a critical aspect of chemical reactions.
Equilibrium, in the context of chemistry, refers to a state where the rates of the forward and backward reactions are equal, resulting in no net change in the concentrations of reactants and products over time. This concept was first introduced by Berthollet in 1803, who discovered that some chemical reactions are reversible. Achieving equilibrium is essential because it signifies a stable state where the reaction has reached a balance, with no observable changes in the system's properties.
The rate equation serves as a tool to predict the relationship between reaction rates and the concentrations of reactants and products. This prediction is experimentally determined and provides valuable insights into the behaviour of chemical reactions. However, it's important to note that catalysts, while influencing reaction rates, do not appear in the balanced overall chemical equation. This is because catalysts do not affect the equilibrium constant, as they impact both the forward and backward reactions equally.
Despite not appearing in the equation, catalysts play a crucial role in establishing equilibrium. They speed up the rate at which equilibrium is reached, making it faster to attain a stable state. This is particularly relevant in dynamic equilibrium, where the rates of the forward and backward reactions are equal, but changes still occur at the molecular level. The presence of a catalyst does not change the equilibrium constant but influences the rate at which it is reached.
In conclusion, while the rate equation is essential for understanding reaction rates, its role in establishing equilibrium conditions is equally, if not more, important. By predicting the relationship between reaction rates and concentrations, the rate equation provides a foundation for comprehending the dynamic nature of chemical reactions and their journey towards equilibrium.
Medical Marijuana and Law Enforcement: Who's Eligible?
You may want to see also

The rate law is experimentally determined
The rate law provides a relationship between reaction rates and reactant concentrations. The units of the rate constants for zero, first, second, and nth-order reactions are tabulated. Differential rate laws are used to express the rate of a reaction in terms of the change in the concentration of reactants over a small interval of time. Ordinary rate law is a differential rate equation since it offers insight into the instantaneous rate of the reaction. Integrated rate equations express the concentration of the reactants in a chemical reaction as a function of time.
To determine a rate law, we need to find the values of the exponents n, m, and p, and the value of the rate constant, k. If we are given the reaction orders for a reaction, we have the values of the coefficients we need to write the rate law. For example, if we are told that a reaction is second order in A, we know that n is equal to 2 in the rate law. If we are given data from two or more experiments at the same temperature with different concentrations of reactants and different rates, we can determine the exponents in the differential rate law for the reaction.
Reactions are often monitored by some form of spectroscopy, where "light" or some other frequency of electromagnetic radiation passes through a reaction sample. The light interacts with the molecules in the sample, which absorb particular frequencies of light. Less light exits the sample than entered it; the amount that exits is measured by a detector on the other side. The rate equation is generally created on an empirical basis to establish a relationship between the rate and reactant concentration. The dependence on catalysis concentration is generally complex and is not needed in most cases. The rate constant depends on the particular concentration of any active catalyst that is involved.
Beer Law: Can It Handle Higher Concentrations?
You may want to see also

The rate equation is based on empirical data
The rate equation, or rate law, is a mathematical expression that describes the relationship between the rate of a chemical reaction and the concentration of its reactants. The rate equation is experimentally determined and can be used to predict the relationship between the rate of a reaction and the concentrations of reactants and products. It is based on empirical data, with the rate equation for an elementary reaction being empirically found to obey the law of mass action. This law predicts that the rate depends only on the concentrations of the reactants, raised to the powers of their stoichiometric coefficients.
The rate equation is derived from experimental data using the method of initial rates to determine reaction orders, the rate constant, and the rate law for a reaction. The rate law can be determined by selecting two sets of rate data that differ in the concentration of only one reactant and setting up a ratio of the two rates and the two rate laws. After canceling terms that are equal, an equation is obtained that contains only one unknown, the coefficient of the concentration that varies. This equation can then be solved for the coefficient.
The rate equation is expressed as:
> {\displaystyle k} is the reaction rate constant or rate coefficient and at very few places velocity constant or specific rate of reaction. Its value may depend on conditions such as temperature, ionic strength, surface area of an adsorbent, or light irradiation. If the reaction goes to completion, the rate equation for the reaction rate ... {\displaystyle v\;=\;k [\ce {A}]^{x} [\ce {B}]^{y}} applies throughout the course of the reaction.
The rate equation is generally created on an empirical basis to establish a relationship between the rate and reactant concentration. The dependence on catalyst concentration is generally complex and, in most cases, not needed. Catalysts can influence the rate of reaction, but they do not appear in the balanced overall chemical equation. The rate equation may, however, include the concentration of active particles on the surface of the catalyst.
In conclusion, the rate equation is based on empirical data, with the rate law being experimentally determined and used to predict the relationship between reaction rate and reactant concentrations. The rate equation is derived from empirical data using the method of initial rates, and it describes the relationship between the rate of a chemical reaction and the concentration of its reactants.
Impeachment and Lawmaking: Can a President Still Legislate?
You may want to see also

The concentration of active particles on a catalyst's surface should be included in the rate equation
The rate of a chemical reaction is influenced by the concentrations of the substances involved. The rate law is used to predict the relationship between the rate of a reaction and the concentrations of reactants and products. The rate equation is generally created on an empirical basis to establish this relationship.
Catalysts can influence the rate of a reaction, and their concentration may be included in the rate equation. However, the dependence of the rate on catalyst concentration is often complex and not always required. In some cases, the catalyst concentration is less than that of the reactants, and it may be challenging to model the interaction between the catalyst and reactants.
For heterogeneous catalysts, which are not part of the reaction mixture, the concentration in the mixture is defined as zero. However, the concentration of active particles on the surface of a heterogeneous catalyst should be included in the rate equation. This is because the rate of the reaction is proportional to the surface area of the catalyst, and a higher concentration of active particles on the catalyst's surface can increase the rate of the reaction.
Additionally, the rate of a reaction can be influenced by factors such as temperature, pressure, and the presence of a catalyst. These factors are observed in the change of the rate constant, K, of the reaction.
Amnesty Laws: Freedom or Confinement?
You may want to see also
Frequently asked questions
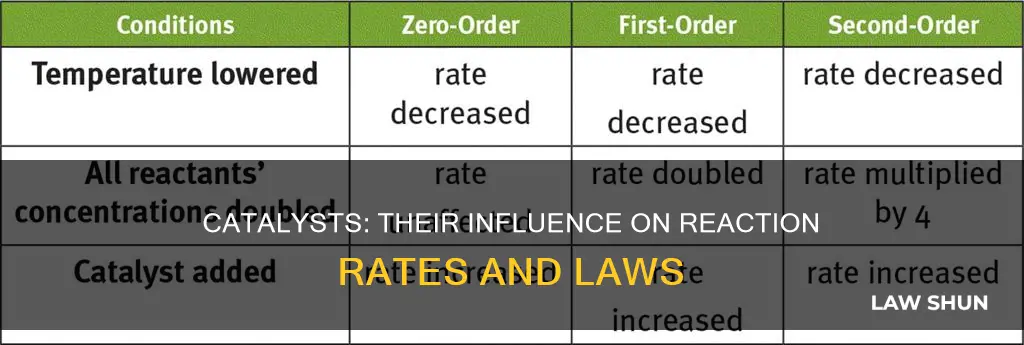
Yes, catalysts can appear in the rate law. The rate law is experimentally determined and can be used to predict the relationship between the rate of a reaction and the concentrations of reactants and products.
Catalysts can influence the rate of reaction by lowering the activation energy required for the reaction to occur. This results in an increased number of reactant molecules with sufficient energy to undergo a reaction, thereby increasing the rate of reaction.
Catalysts are not consumed or produced during the reaction, so their concentration remains constant throughout. Therefore, they do not appear in the balanced overall chemical equation.
The presence of a catalyst can make the reaction multi-step and more complicated. The rate equation becomes more challenging to model as it involves a three (or more) particle interaction.







