Charles's Law, also known as the law of volumes, is an experimental gas law formulated by French physicist Jacques Charles in the 1780s. The law describes the relationship between the volume of a gas and its temperature when pressure is kept constant. While the law is often used to explain how gases expand when heated, it also provides insight into how a decrease in temperature leads to a decrease in volume. Charles's Law is expressed in terms of absolute temperature, which is measured using the Kelvin scale. This is because the Kelvin scale defines absolute zero as the complete cessation of molecular motion, corresponding to -273.15°C or -459.67°F. As a result, Charles's Law cannot be expressed in degrees Celsius, as the Celsius scale does not account for this critical temperature threshold.
| Characteristics | Values |
|---|---|
| Can Charles's Law be in Celsius? | No, the Kelvin scale must be used because zero on the Kelvin scale corresponds to a complete stop of molecular motion. |
| What is Charles's Law? | An experimental gas law that describes how gases tend to expand when heated. |
| Who is it named after? | French physicist Jacques Charles (1746-1823) |
| What does it state? | The volume of a given mass of gas varies directly with the absolute temperature of the gas when pressure is kept constant. |
| What is the absolute temperature? | Temperature measured with the Kelvin scale. |
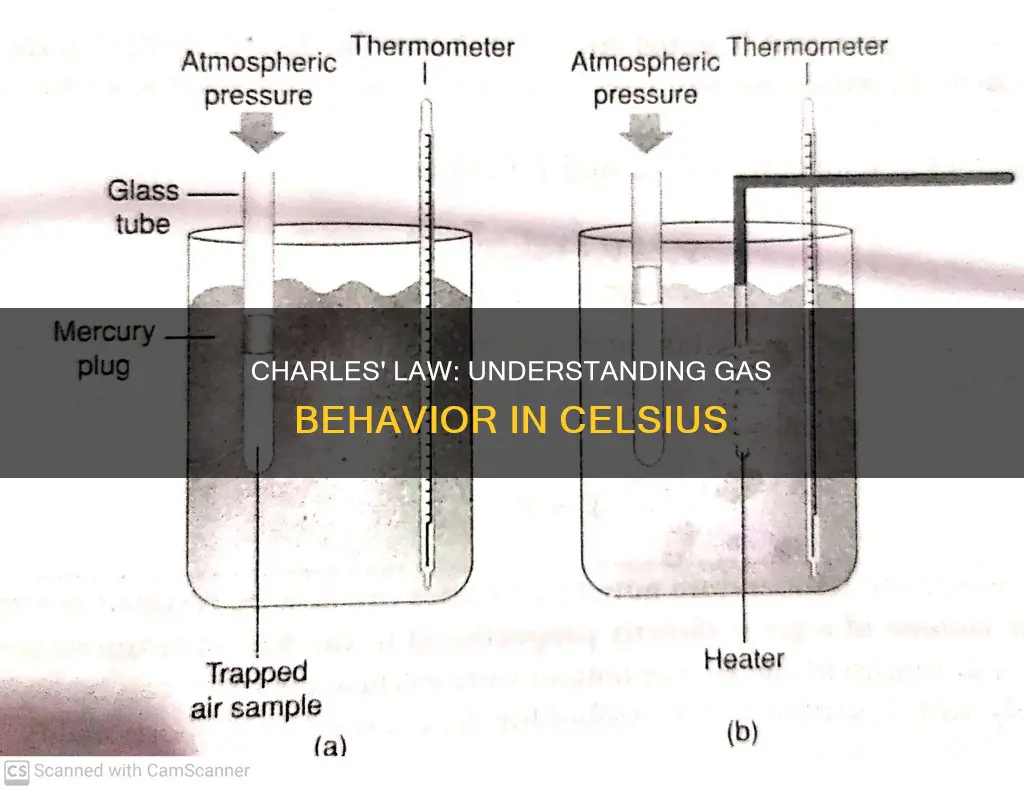
| What happens when a gas is heated? | Its molecules increase in kinetic energy and push the movable piston outward, resulting in an increase in volume. |
| What happens when the temperature decreases? | A decrease in volume. |
| What is the lowest possible temperature according to Charles's Law? | -273°C |
What You'll Learn
- Charles's Law is based on the work of French physicist Jacques Charles
- The law states that the volume of gas varies directly with temperature when pressure is kept constant
- Absolute temperature is measured with the Kelvin scale
- Temperatures in Celsius will not work for Charles's Law calculations
- The lowest possible temperature according to the law is -273°C

Charles's Law is based on the work of French physicist Jacques Charles
Charles's Law, also known as the law of volumes, is an experimental gas law that describes how gases tend to expand when heated. It was named after French physicist Jacques Charles, who formulated the original law in his unpublished work from the 1780s. Charles's Law states that the volume of a given mass of gas varies directly with the absolute temperature of the gas when pressure is kept constant. The absolute temperature is the temperature measured with the Kelvin scale. The Kelvin scale must be used because zero on the Kelvin scale corresponds to a complete stoppage of molecular motion.
The mathematical relationship of Charles's Law can be represented by the following equation: V1/T1 = V2/T2, where V1 and T1 stand for the initial volume and temperature of a gas, while V2 and T2 stand for the final volume and temperature. This equation can be used to calculate any one of the four quantities if the other three are known. It is important to note that the direct relationship will only hold if the temperatures are expressed in Kelvin. Temperatures in Celsius will not work.
The French natural philosopher Joseph Louis Gay-Lussac confirmed the discovery in a presentation to the French National Institute on 31 January 1802, although he credited the discovery to unpublished work from the 1780s by Jacques Charles. John Dalton, in a series of four essays presented between 2 and 30 October 1801, demonstrated through experiments that all the gases and vapours he studied expanded by the same amount between two fixed points of temperature.
Charles's Law can be derived from the kinetic theory of gases, which relates the macroscopic properties of gases, such as pressure and volume, to the microscopic properties of the molecules that make up the gas, particularly their mass and speed. To derive Charles's Law from kinetic theory, a microscopic definition of temperature is necessary, which can be taken as the temperature being proportional to the average kinetic energy of the gas molecules.
How Can a Woman Get Alimony in Nevada?
You may want to see also

The law states that the volume of gas varies directly with temperature when pressure is kept constant
Charles's Law, also known as the law of volumes, is an experimental gas law that describes how gases tend to expand when heated. The law was named after French physicist Jacques Charles, who formulated the original law in his unpublished work from the 1780s. The French natural philosopher Joseph Louis Gay-Lussac confirmed the discovery in a presentation in 1802, crediting Charles.
The law states that the volume of a given mass of gas varies directly with the absolute temperature of the gas when pressure is kept constant. In other words, when the pressure on a sample of a dry gas is held constant, the Kelvin temperature and the volume will be in direct proportion. This relationship can be expressed mathematically as:
> V1/T1 = V2/T2
Where V1 and T1 represent the initial volume and temperature of a gas, and V2 and T2 represent the final volume and temperature. This equation can be used to calculate any one of the four quantities if the other three are known.
It is important to note that the direct relationship between volume and temperature in Charles's Law only holds if the temperatures are expressed in Kelvin. This is because zero on the Kelvin scale corresponds to a complete stoppage of molecular motion, which is not the case with Celsius. Therefore, Charles's Law cannot be used with temperatures in Celsius. The relationship between the Celsius and Kelvin scales can be expressed as:
> K = °C + 273
Gold Dot Ammunition: Available to Both Civilians and Law Enforcement
You may want to see also

Absolute temperature is measured with the Kelvin scale
The Kelvin scale is used to measure absolute temperature because it is the only scale on which zero corresponds to a complete stoppage of molecular motion. This is important because Charles's Law, which describes the relationship between the volume of a gas and its temperature, assumes that the volume of a gas will decrease to zero as the temperature decreases to absolute zero. Charles's Law states that the volume of a given mass of gas varies directly with the absolute temperature of the gas when pressure is kept constant. In other words, as the Kelvin temperature increases, the volume of the gas increases, and vice versa.
The direct relationship between volume and temperature in Charles's Law will only hold if the temperatures are expressed in Kelvin. Temperatures in Celsius will not work because the Celsius scale does not have a true zero point; it is based on the melting and boiling points of water, which are not absolute temperatures. To convert a temperature in degrees Celsius to Kelvin, one can use the formula K = °C + 273. For example, the triple point of water, which is defined as exactly 0.01 °C, is 273.16 Kelvin.
The Kelvin scale was formally added to the International System of Units in 1954, and it has been revised over time as our understanding of thermal physics has improved. For example, the 2019 revision of the SI defined the Kelvin in terms of energy, setting the Boltzmann constant to exactly 1.380649×10^23 joules per Kelvin.
Laws Rescinded: Can Any Legislation Be Reversed?
You may want to see also

Temperatures in Celsius will not work for Charles's Law calculations
Charles's Law, also known as the law of volumes, is an experimental gas law that describes how gases tend to expand when heated. The law was formulated by French physicist Jacques Charles in the 1780s. It states that the volume of a given mass of gas varies directly with the absolute temperature of the gas when the pressure is kept constant.
The absolute temperature in Charles's Law is the temperature measured with the Kelvin scale. The Kelvin scale is used because zero on the Kelvin scale corresponds to a complete stop of molecular motion. The direct relationship of Charles's Law can be represented mathematically as:
> > \[\dfrac{V_1}{T_1} = \dfrac{V_2}{T_2} \nonumber \]
This equation can be used to calculate any one of the four quantities if the other three are known. However, this direct relationship will only hold if the temperatures are expressed in Kelvin. Temperatures in Celsius will not work for Charles's Law calculations. This is because a gas at 10 Kelvin has only half the heat energy of a gas at 20 Kelvin, which is not true for a gas at 10 degrees Celsius compared to 20 degrees Celsius.
The relationship between the Kelvin and Celsius scales can be expressed as:
> > \(\text{K} = \: ^\text{o} \text{C} + 273\)
Lien Laws: Can Attorneys and Law Firms Claim?
You may want to see also

The lowest possible temperature according to the law is -273°C
Charles's Law, also known as the law of volumes, is an experimental gas law that describes how gases tend to expand when heated. It states that the volume of a given mass of gas varies directly with the absolute temperature of the gas when pressure is kept constant. The absolute temperature is measured with the Kelvin scale, where zero on the Kelvin scale corresponds to a complete stoppage of molecular motion.
Charles's Law can be used to compare changing conditions for a gas. The mathematical relationship of the law can be used to calculate any one of four quantities (initial volume, final volume, initial temperature, and final temperature) if the other three are known. It is important to note that the temperatures in this law must be expressed in Kelvin, not Celsius.
According to Charles's Law, the lowest possible temperature is -273°C. At this temperature, the volume of any gas is equal to zero. This is because the volume would be negative below this temperature, which is not possible. This temperature of -273°C is known as "absolute zero" and was originally defined in terms of the second law of thermodynamics. It is important to note that Thomson, who described this law in 1852, did not assume that this was equal to the "zero-volume point" of Charles's Law but rather stated that it provided the minimum temperature that could be attained.
Common-Law Spouses: VA Benefits Eligibility
You may want to see also
Frequently asked questions
No, Charles's Law can only be used with temperatures expressed in Kelvin. This is because zero on the Kelvin scale corresponds to a complete stoppage of molecular motion.
Charles's Law (or the law of volumes) is an experimental gas law that describes how gases tend to expand when heated. It states that the volume of a given mass of gas varies directly with the absolute temperature of the gas when pressure is kept constant.
The mathematical relationship of Charles's Law is:
> > \[\dfrac{V_1}{T_1} = \dfrac{V_2}{T_2} \nonumber\]
>
This equation can be used to calculate any one of the four quantities if the other three are known.
The lowest possible temperature is -273°C or -266.66°C according to Gay-Lussac's figures.