Coulomb's law describes the interaction between charged particles, and it can be used to explain the solubility of ionic compounds in water. Ionic compounds often have some degree of solubility in water, and the solubility of different ionic compounds can vary. Coulomb's law can help explain these variations in solubility by considering the attraction between ions and the attraction between ions and water molecules. The law is dependent on the properties of the intervening medium and holds for stationary point charges. It can be applied when the inverse square law is obeyed, and it describes the electrostatic force between two charges, which always acts along the line joining them.
| Characteristics | Values |
|---|---|
| Coulomb's Law | Describes the interaction between ions (charged particles) |
| Holds good for stationary point charges | |
| Can be applied when the inverse square law is obeyed | |
| Governs the attraction of water to ions | |
| Describes the interaction between mobile ions in aqueous solutions of ionic substances | |
| Solubility | Ionic compounds often have some solubility in water |
| All nitrate salts are soluble | |
| Water has a large dielectric constant, which reduces the electrostatic interaction between particles |
What You'll Learn
- Coulomb's Law describes the interaction between charged particles
- The law is dependent on the properties of the intervening medium
- It can be used to predict varying degrees of solubility in water
- The dielectric constant of the solvent reduces electrostatic interaction
- Coulomb's Law is not universal and only applies to stationary charges

Coulomb's Law describes the interaction between charged particles
Coulomb's law, an experimental law of physics, describes the interaction between charged particles. It calculates the amount of force between two electrically charged particles at rest. This force is conventionally called the electrostatic force or Coulomb force. The law was first published in 1785 by French physicist Charles-Augustin de Coulomb, who used a torsion balance to study the repulsion and attraction forces of charged particles.
The force of attraction or repulsion between two charged bodies is directly proportional to the product of their charges and inversely proportional to the square of the distance between them. Coulomb's law can be applied to describe the repulsive force between two electrons, although it does not fully account for the interplay between multiple electrons and nuclei in multi-electron atoms. It is limited to the description of a two-particle system.
The electrostatic force between two point charges always acts along the straight line joining the two charges. It can be used to gain insight into the form of the magnetic field generated by moving charges. Coulomb's law can be expanded to moving test particles, with the assumption that the charge is invariant to the observer.
In the context of solubility, Coulomb's law describes the interaction between ions (charged particles). When two aqueous solutions of ionic substances are mixed, the mobile ions in each solution interact with each other. The dielectric constant of the solvent, such as water, plays a role in reducing the electrostatic interaction between the particles. When the charges have the same sign, they repel each other, and when they have opposite signs, they are attracted to each other.
Congress' Power: Can They Control Speed Limits?
You may want to see also

The law is dependent on the properties of the intervening medium
Coulomb's law describes the interaction between ions (charged particles). It holds for stationary point charges and is dependent on the properties of the intervening medium. This means that the law is not universal and is influenced by the nature of the medium separating the charges.
The force between two charges depends on the intervening medium, whereas the gravitational force is independent of it. Coulomb's law can be applied when the inverse square law is obeyed. It becomes difficult to implement when charges are of arbitrary shape, as the distance between the charges cannot be determined.
In the context of solubility, Coulomb's law helps explain the interaction between ions and water molecules. For example, when ions of opposite charge encounter each other, the attractive force between them can overcome the interaction with water molecules, leading to the formation of a compound that is not soluble in water.
Additionally, the size of the ions plays a role in solubility. Smaller ions are closer together, making it harder for water molecules to pull them apart. This concept can be understood using Coulomb's law, which considers the electrostatic force between charges.
Coulomb's law also accounts for the dielectric constant of the solvent, which affects the electrostatic interaction between particles. Water, for instance, has a large dielectric constant, reducing the electrostatic interaction between particles.
Congressional Power: Revising Laws
You may want to see also

It can be used to predict varying degrees of solubility in water
Coulomb's law describes the interaction between ions (charged particles). It can be used to predict varying degrees of solubility in water. This is because the solubility of ionic compounds in water depends on the attraction between the ions and the water molecules. Smaller ions, for example, are held more tightly by water molecules, making it harder for them to be dissolved. Coulomb's law can be used to understand these interactions and predict the solubility of different ionic compounds.
Coulomb's law, however, is not a universal law and has some limitations. It only applies to stationary point charges and depends on the properties of the intervening medium. It is difficult to apply in cases where charges are of arbitrary shape since the distance between charges cannot be determined. For example, Coulomb's law cannot be used to directly calculate the charge on large planets.
In the context of solubility, Coulomb's law can be used to understand the interactions between ions and water molecules. When an ionic solid dissolves in water, the individual ions interact with the water molecules. The strength of this interaction depends on the charges of the ions and the polarity of the water molecules. Water has a large dielectric constant, which reduces the electrostatic interaction between particles.
The solubility of an ionic compound in water is influenced by the balance between the attraction of the ions for each other and the attraction of the ions for the water molecules. When ions of opposite charge encounter each other, they are attracted and can form a compound that is not soluble in water. This compound falls out of the solution and is called a precipitate. On the other hand, if the attraction between the ions and the water molecules is stronger, the ions remain dissolved in the water, and the compound is considered soluble.
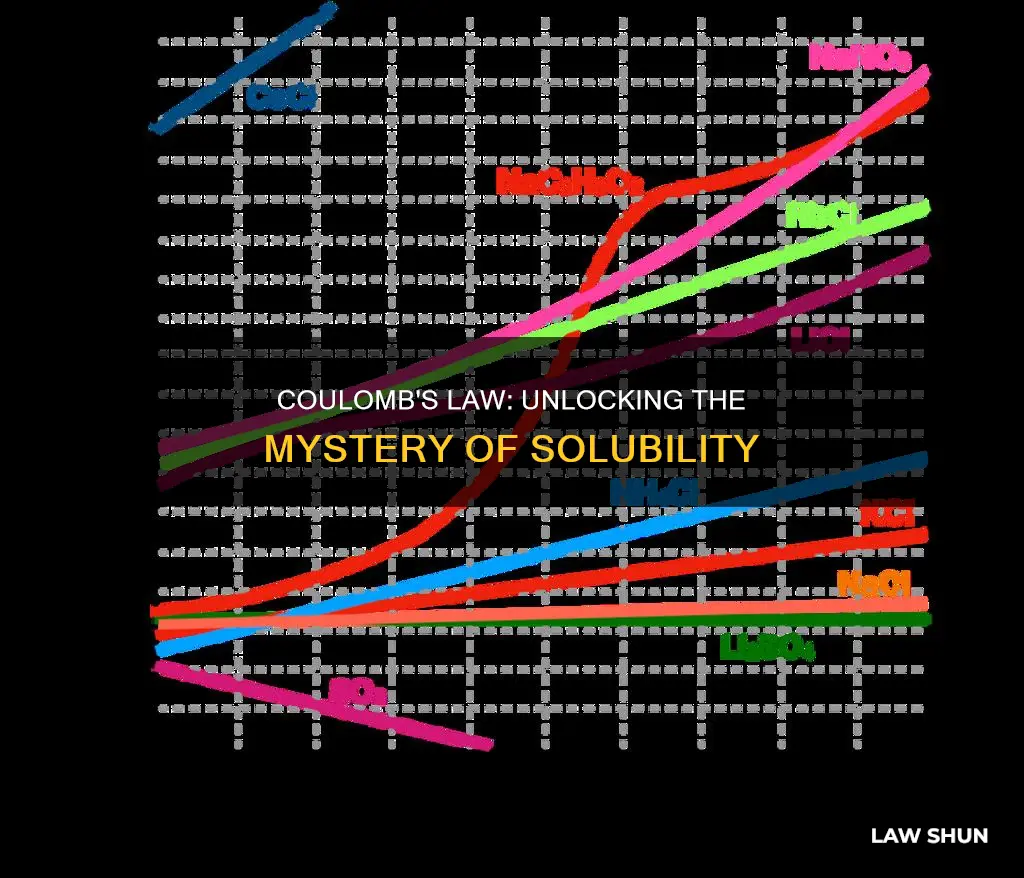
By applying Coulomb's law, it is possible to predict which ionic compounds will be more or less soluble in water. For example, lithium fluoride and magnesium oxide contain cations and anions of similar sizes, but lithium fluoride is much more soluble in water at room temperature. This can be explained by considering the Coulombic attraction between the ions and how they interact with water molecules.
City Council Powers: Lawmaking and Enforcement Explored
You may want to see also

The dielectric constant of the solvent reduces electrostatic interaction
Coulomb's law describes the interaction between ions (charged particles) in a solution. The dielectric constant of the solvent is an important factor in understanding solubility. It influences the electrostatic interactions between charged particles.
The dielectric constant of a solvent measures its ability to resist an electric current. A higher dielectric constant indicates a stronger resistance to electric current. This is an important property because it affects the behaviour of charged particles within the solvent. When the solvent has a high dielectric constant, it weakens the electrostatic force between charged particles.
In the context of solubility, the dielectric constant of the solvent plays a crucial role in determining how ions interact. For example, in an aqueous solution, water has a large dielectric constant. This high value reduces the electrostatic interaction between particles. When the charges of the ions are the same, they repel each other (F > 0). Conversely, when the charges are opposite, they are attracted to each other (F < 0). The dielectric constant of the solvent influences the strength of these attractive and repulsive forces.
The effect of the dielectric constant on electrostatic interactions is not limited to aqueous solutions. Studies have investigated the impact of solvent dielectric constants on the behaviour of charged nanoparticles. It was found that a solvent with a higher dielectric constant can enhance the electrostatic repulsion between nanoparticles, making them less likely to coalesce. This effect is not only dependent on the value of the dielectric constant but also on the nature of the solvent and the specific ions involved.
The influence of the solvent's dielectric constant on electrostatic interactions is a complex phenomenon. It depends on various factors, including the distance between charges, the solvent's permittivity, and the heterogeneity of the solvent's dielectric properties. To accurately capture these effects, various theoretical and experimental approaches have been employed, including continuum models, nonlocal electrostatics, and the calculation of generalized susceptibility.
Commission Conundrum: Minimum Pay Law Mystery
You may want to see also

Coulomb's Law is not universal and only applies to stationary charges
Coulomb's law describes the interaction between charged particles or ions. It states that the force of attraction or repulsion between two charged bodies is directly proportional to the product of their charges and inversely proportional to the square of the distance between them.
Coulomb's law is a simplification of Maxwell's equations under specific conditions (electrostatic = no moving charges). The universal law is Maxwell's equations, which are more complex and sometimes challenging to solve. Therefore, the simpler Coulomb's law is used when applicable, and the more comprehensive but universal Maxwell's equations are used when necessary.
Gauss's law is also one of Maxwell's equations and is, therefore, universal. Although it can be derived from Coulomb's law in the context of electrostatics, it is also applicable outside of that context. This is because the expression for the electric field reduces to Coulomb's law for non-relativistic speeds of the point charge.
In summary, Coulomb's law is not universal because it only applies to stationary charges. When charges are in motion, other factors, such as magnetic forces, come into play, and more comprehensive laws like Maxwell's equations must be considered.
Why Can Citizens Buy Speer Gold Dot Law Enforcement?
You may want to see also
Frequently asked questions
Coulomb's law describes the interaction between charged particles, or ions, and can be used to explain solubility. It holds for stationary point charges and obeys Newton's third law.
Coulomb's Law can be used to explain the solubility of ions in water. It describes the attraction between the ions and the water. The smaller the ions, the closer they are together, and the harder it is for water molecules to pull the ions away from each other.
Lithium chloride is the least soluble of the lithium salts. However, lithium bromide is more soluble than lithium iodide, which indicates that there are multiple factors influencing solubility. This is because there are two interactions to consider: the attraction of the ions to each other, and the attraction of the ions to the water molecules.
Coulomb's Law is not universal and depends on the properties of the intervening medium. It can only be applied when the inverse square law is obeyed. It is difficult to implement when charges are irregularly shaped as the distance between charges cannot be determined.







